ACR-2000 Urine Albumin-to-Creatinine Ratio (UACR) Analyzer
Compact UACR analyzer for early kidney risk screening in primary healthcare and medical examination settings
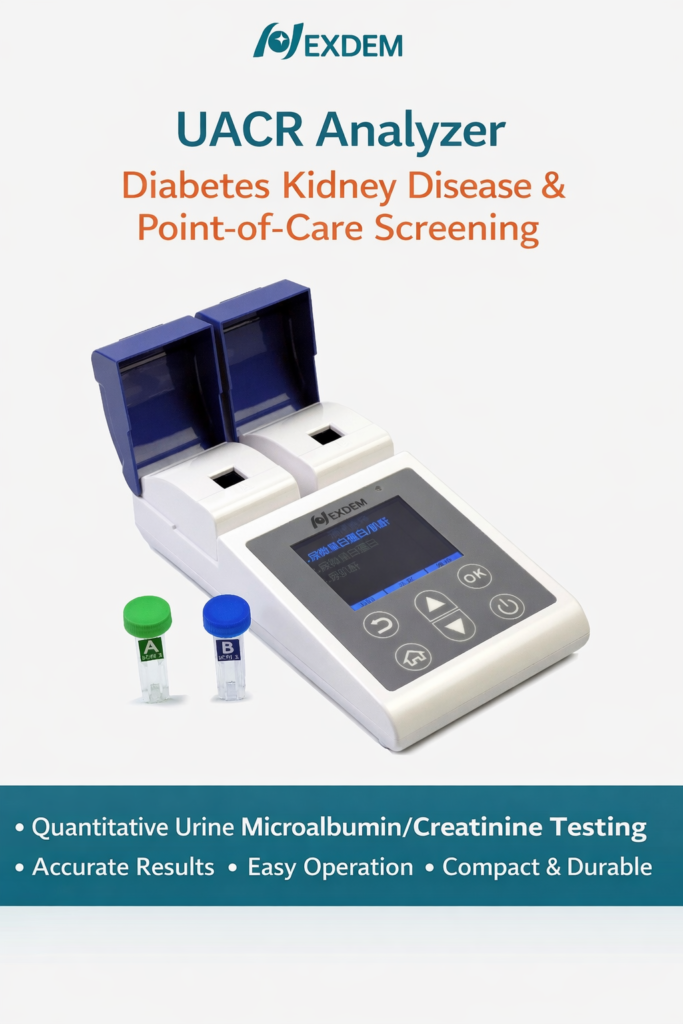
ACR-2000 UACR Analyzer
ACR-2000 UACR Analyzer is a compact and portable urine analysis system designed for accurate urine albumin-to-creatinine ratio (UACR) testing. It is widely applied in primary healthcare institutions, occupational health programs, community clinics, and medical examination centers where early kidney disease screening and routine urine testing are required.
By applying advanced digital spectrophotometric technology, the ACR-2000 UACR Analyzer delivers fast, reliable, and reproducible urine testing results. The system supports standardized screening workflows and early identification of potential kidney-related risks, including diabetes-associated kidney conditions and early-stage chronic kidney disease.
Product Overview
The ACR-2000 UACR Analyzer is engineered specifically for frontline healthcare environments where efficiency, accuracy, and portability are essential. Its compact footprint and lightweight design make it easy to deploy in both fixed clinical settings and mobile screening programs.
The guided operating system allows healthcare professionals to complete UACR testing with minimal training, reducing operational barriers while ensuring consistent and standardized testing performance across different users and locations.
Why UACR Testing Matters in Kidney Health Screening
Urine albumin-to-creatinine ratio (UACR) testing is a clinically recommended indicator for early detection of kidney damage. Compared with conventional urine protein tests, UACR testing provides higher sensitivity for identifying microalbuminuria at an early stage.
The ACR-2000 UACR Analyzer enables reliable UACR measurement even when routine urine tests appear normal, supporting early intervention and long-term kidney disease management in at-risk populations such as patients with diabetes or hypertension.
Application Scenarios of the ACR-2000 UACR Analyzer
The ACR-2000 urine testing system is suitable for a wide range of screening and examination environments, including:
- Kidney health screening programs
- Diabetes-related kidney risk monitoring
- Occupational health examinations
- Community and primary healthcare facilities
- Medical examination and health screening centers
Its portability and fast testing capability make it particularly suitable for high-throughput screening workflows.
Testing Capabilities
Urine Microalbumin Testing
- Measurement range: 5–420 mg/L
- Method: Immunoturbidimetric assay
Urine Creatinine Testing
- Measurement range: 1.0–48.6 mmol/L
- Method: Enzymatic method
UACR Calculation
- Calculation range: 1.0–2200 mg/g
By combining albumin and creatinine testing, the ACR-2000 UACR Analyzer ensures accurate urine albumin-to-creatinine ratio analysis for standardized kidney risk screening.
Performance & Efficiency
- Single-sample test time: approximately 7 minutes
- Batch testing capacity: up to 4 samples within 12 minutes
- Constant-temperature heating system: 37°C ±1°C
- Simultaneous heating of up to 8 cuvettes
These performance features support efficient processing of multiple samples while maintaining analytical consistency in daily screening workflows.
Optical & Analytical System
- Multi-wavelength detection: 415 / 450 / 485 / 520 / 555 / 590 / 625 / 690 nm
- Absorbance linear range: 0–2.0 ABS
- Repeatability: RSD < 0.5%
The advanced optical system ensures reliable and reproducible results suitable for routine urine testing and large-scale screening applications.
Design & Portability
- Dimensions: 205 × 110 × 55 mm
- Weight: approximately 450 g
- Power supply: 12V power adapter
- Built-in rechargeable lithium battery
The lightweight and compact design allows flexible deployment in clinics, mobile screening units, and community-based health programs.
Display, Storage & Connectivity
- Color LCD display: 240 × 160 resolution
- Internal storage: up to 4,000 test records
- USB interface for data export or PC connection
- Supports data management and optional cloud-based upload
These features support efficient data handling and integration into existing screening workflows.
Ease of Use
- Guided user interface with step-by-step operation prompts
- Minimal training required for routine operation
- Designed for consistent and repeatable daily screening workflows
Regulatory & Compliance
- Medical device registration number: 20192220373
- Designed in compliance with relevant national regulations and industry standards
Service & Support
- Installation and operational training
- Technical support services
- Minimum one-year warranty
Request More Information
Looking for a reliable ACR-2000 UACR Analyzer to support urine testing, kidney risk screening, and preventive healthcare programs?
Contact us to learn more about product specifications, application support, and integration options.
